  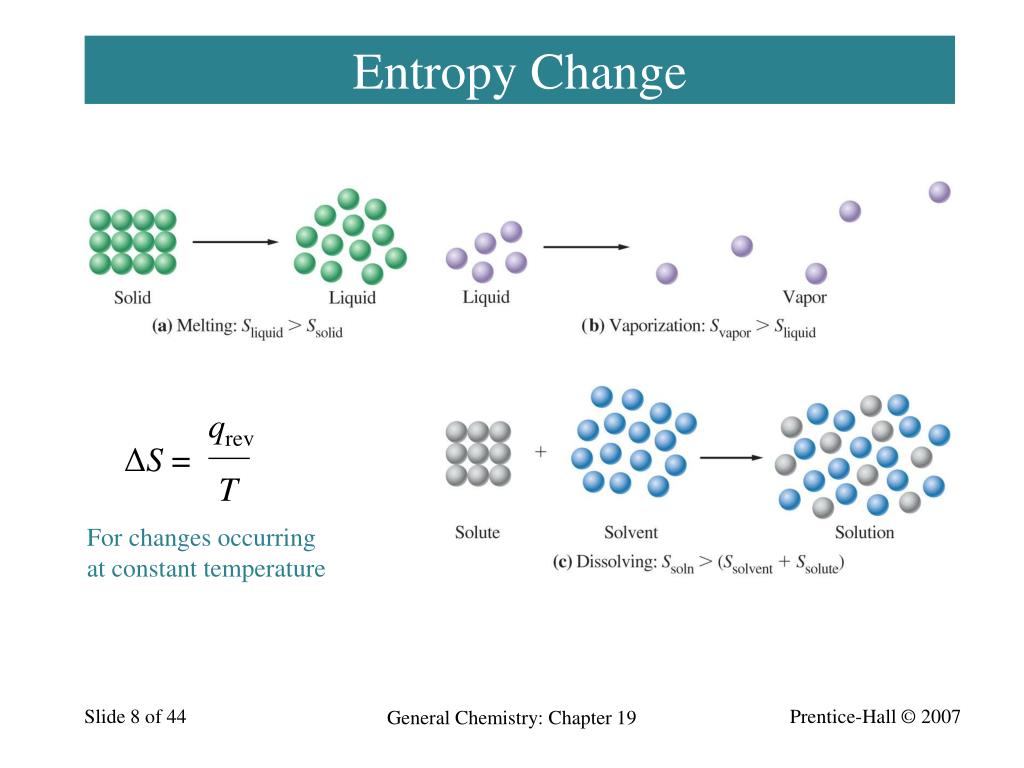
Stars are hotter than planets, for example, which are warmer than icy asteroids, which are warmer still than the vacuum of the space between them. The distribution of the energy is affected by the increase. Although it may have seemed disorderly, and therefore, superficially entropic, in fact, there was enormous potential energy available to do work-all the future energy in the universe.Īs the universe matured, temperature differences arose, which created more opportunity for work. VIDEO ANSWER: As temperature increases, we cause the molecule or atoms to move with more energy. Gravity played a vital role in the young universe. In the early, energetic universe, all matter and energy were easily interchangeable and identical in nature. 1 Heat And Phase Changes Answers Pdf Recognizing the. H U+ (PV) qrev + wrev + (P V) where: qrev and wrev are the most efficient (reversible) heat flow and work, respectively. If a system is thermodynamically favorable each factor (enthalpy temperature and entropy) play a role. Heat Death of the Universe: An Overdose of Entropy The second law of thermodynamics states that the total entropy of a system either increases or remains constant in any spontaneous process it never decreases. 189 heat capacity approaches zero, 187 Carathodory, 295 Clausius statement, 42 Kelvin-Planck statement, 43 principle of entropy increase, 56. Starting from the first law of thermodynamics and the relationship of enthalpy H to internal energy U: U qrev +wrev. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
